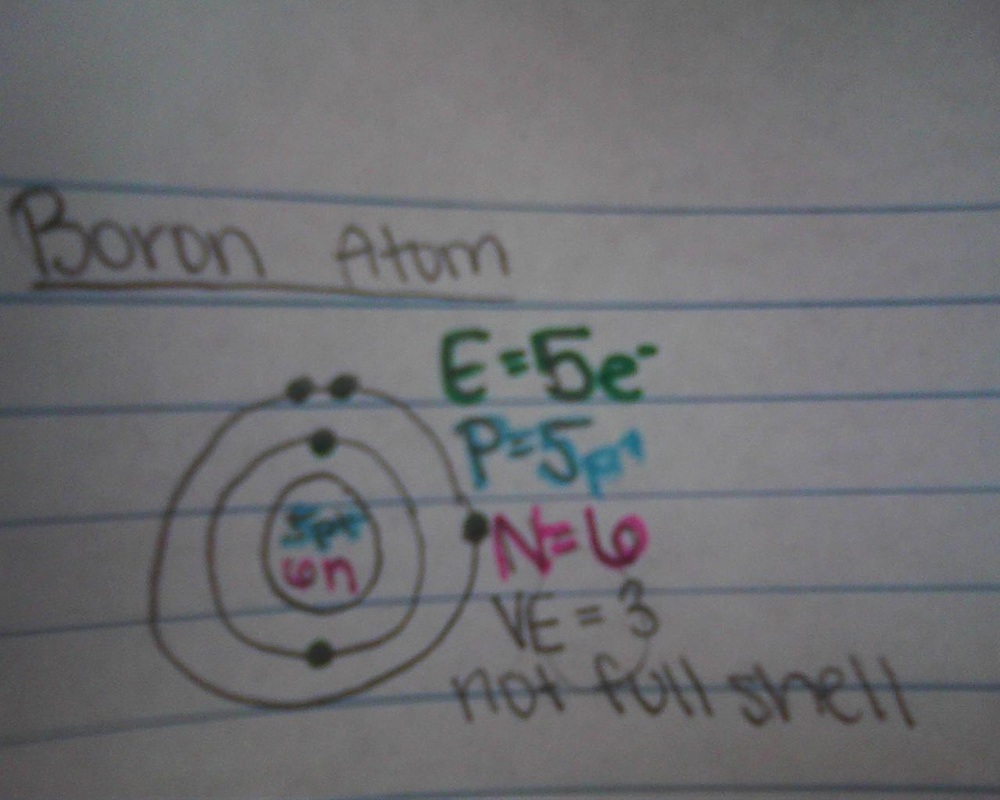
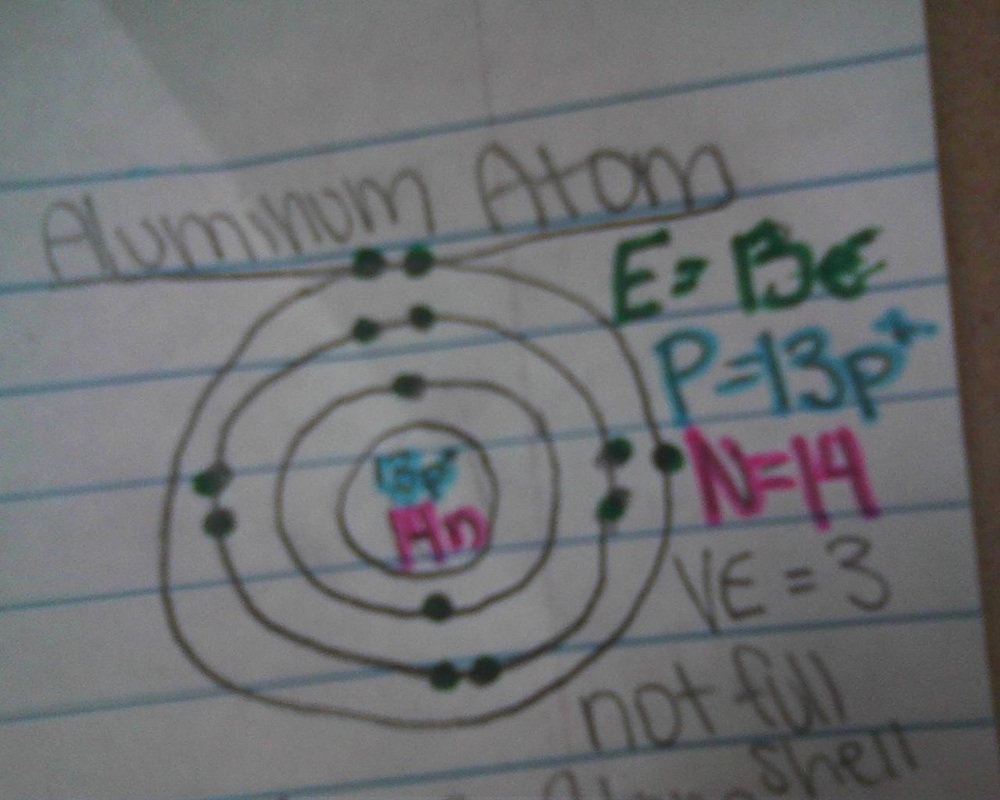
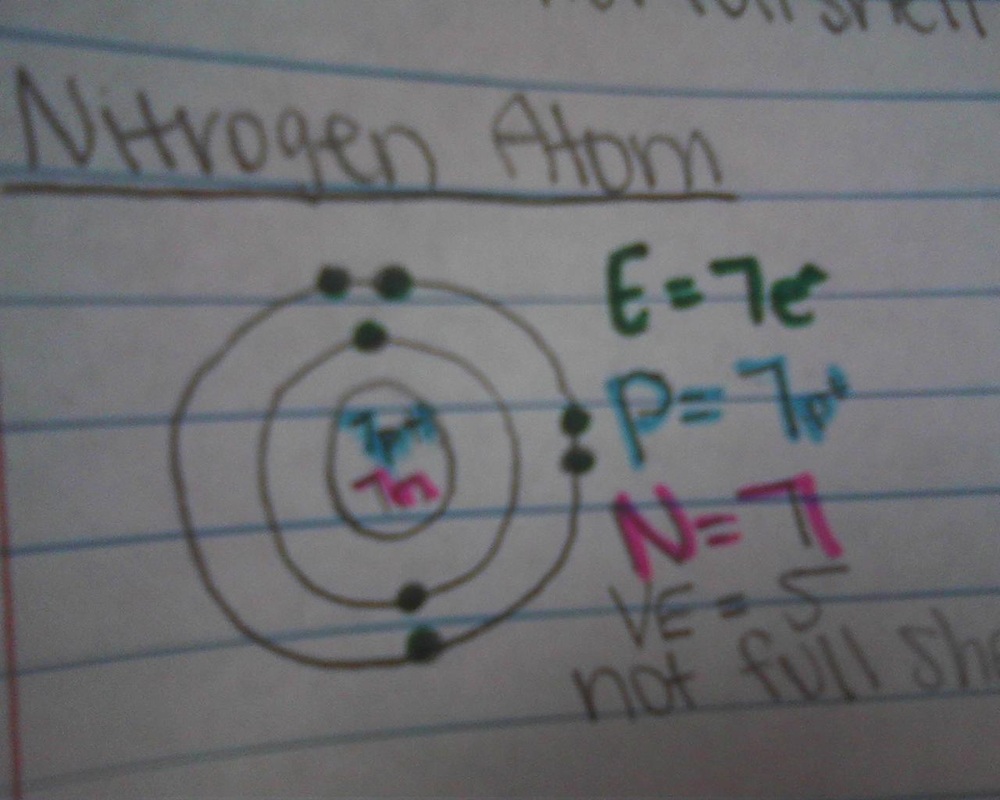
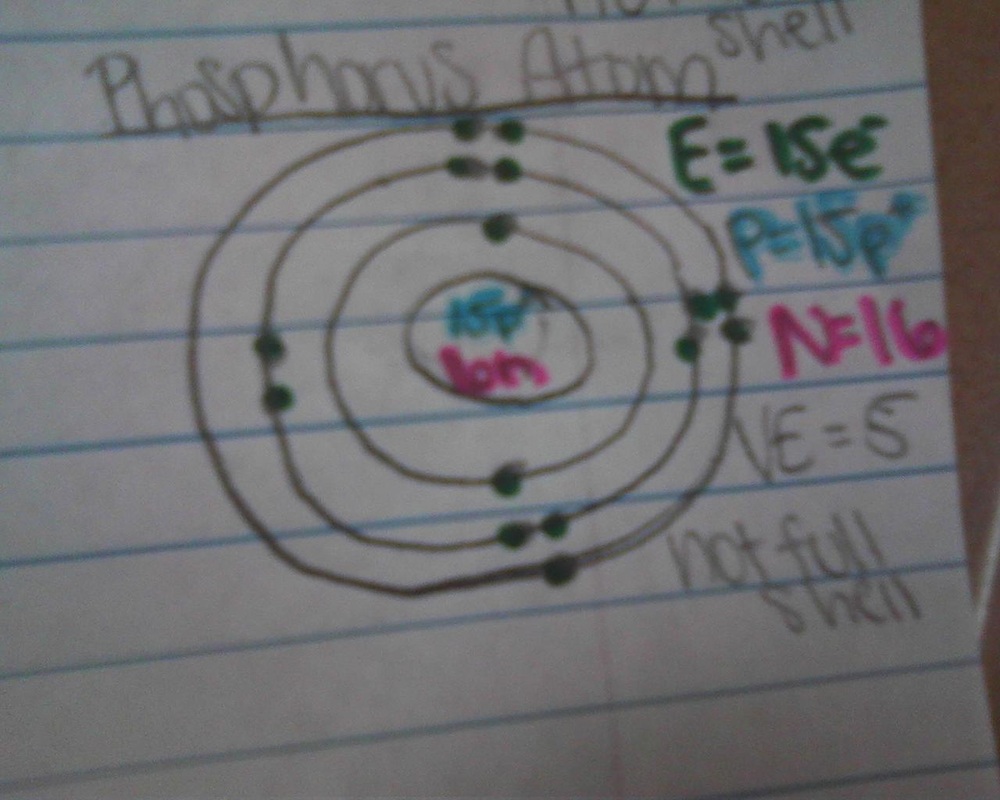
valence electrons
Na is a +1 charge combined with Cl is a -1 charge = table salt
- The electron cloud of an atom is broken up into 7 layers
- the layers are called energy levels or shells
- up to 2 electrons on level one, and up to 8 on every level that follows
- the number of electrons in the outer layer controls how or if atoms react with other atoms
- Valence shell: the outer most energy level of an atom
- Valence electrons: the amount of electrons on the outside energy level
- Atoms combine so that their valence shells are full of electrons
- if a neutral atom's valence shell is full, then the atom will not react with other atoms
- if a neutral atom's valence shell has space available, then the atom will react with other atoms based on the number of electrons found in the valence shell
- some atoms gain electrons to become negative ions
- some atoms lose electrons to become positive ions
- some atoms share electrons to