Vinegar and baking soda lab
Problem: Will increasing the amount of vinegar and baking soda decrease the amount of time it takes for the bag to inflate and explode?
Hypothesis: I think that the more baking soda and vinegar you add, the faster the bag will inflate and explode.
ID: time to explode
IV: amount of baking soda and vinegar
Research:
http://www.rd.com/slideshows/5-best-baking-soda-and-vinegar-cleaning-solutions/
Materials:
Hypothesis: I think that the more baking soda and vinegar you add, the faster the bag will inflate and explode.
ID: time to explode
IV: amount of baking soda and vinegar
Research:
- Hydrogen is released
- becomes full valence shell
- chemical reaction
- should have bubbles
http://www.rd.com/slideshows/5-best-baking-soda-and-vinegar-cleaning-solutions/
Materials:
- vinegar
- baking soda
- zip lock plastic bags
- measuring spoons
- paper
- outside area
- Measure out 6 tablespoons of vinegar and baking soda and place in separate containers
- place the ingredients in bag and quickly seal
- start timer and place bag on floor and watch as it expands
- stand back as the bag explodes
- repeat steps with 12 tablespoons of vinegar and baking soda
CER:
C: The bag with 12 tablespoons of vinegar and baking soda inflated and exploded the fastest, going along with my hypothesis.
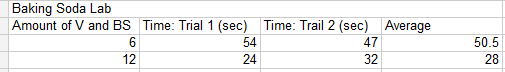
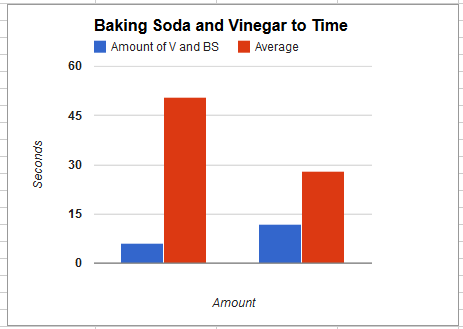
E: In the experiment, the bag with 6 tablespoons of baking soda and vinegar had explosion times of 54 seconds in the first trial and 47 seconds in the second trial with an average of 50.5 seconds. The bag with 12 tablespoons of baking soda and vinegar had explosion times of 24 seconds in the first trial and 32 seconds in the second trial with an average of 28 seconds.
R: During the chemical reaction between vinegar and baking soda carbon dioxide is released. So, the more vinegar and baking soda, the bigger the chemical reaction and therefore more carbon dioxide is released. When more carbon dioxide is released faster, the bag would then inflate and explode faster.
C: The bag with 12 tablespoons of vinegar and baking soda inflated and exploded the fastest, going along with my hypothesis.
E: In the experiment, the bag with 6 tablespoons of baking soda and vinegar had explosion times of 54 seconds in the first trial and 47 seconds in the second trial with an average of 50.5 seconds. The bag with 12 tablespoons of baking soda and vinegar had explosion times of 24 seconds in the first trial and 32 seconds in the second trial with an average of 28 seconds.
R: During the chemical reaction between vinegar and baking soda carbon dioxide is released. So, the more vinegar and baking soda, the bigger the chemical reaction and therefore more carbon dioxide is released. When more carbon dioxide is released faster, the bag would then inflate and explode faster.